September 6, 2006
FDA Ruling Puts Pharmacists in Crossfire
by Daniel C. Vock
Stateline.org
The latest fireworks over the "morning-after pill"
weren't in Congress, or at the U.S. Food and Drug Administration, but in
After months of controversy and a flood of 16,000 comments,
the board followed Gov. Christine Gregoire's (D)
suggestions and refused to give legal protections to pharmacists who for moral
reasons object to dispensing the high-dose birth control pill.
The action, though, doesn't end the tempest in
As
"What this does is shift the burden from doctors with
prescribing rights and privileges to pharmacists," said Deirdre McQuade, the U.S. Conference of Catholic Bishops'
spokeswoman on birth control matters.
"We had been putting our efforts into preventing the
FDA from doing this. … Now it seems like it's going to be a state-based matter,"
McQuade said. She added, however, that the conference
hadn't yet decided what action to take at the state level.
Emergency contraception is controversial because it poses
moral questions similar to those raised in the abortion debate.
The Catholic bishops and others who believe life begins at
conception object to Plan B because of the possibility that it may prevent a
fertilized egg from implanting on the uterine wall.
But those who want greater access to the morning-after pill
argue that the drug works the same as standard oral contraceptives, primarily
by preventing fertilization of a woman's egg. Taken within 72 hours of
unprotected sex, the pill can prevent 89 percent of pregnancies, according to
Plan B's manufacturer, Duramed, a subsidiary of Barr
Pharmaceuticals Inc.
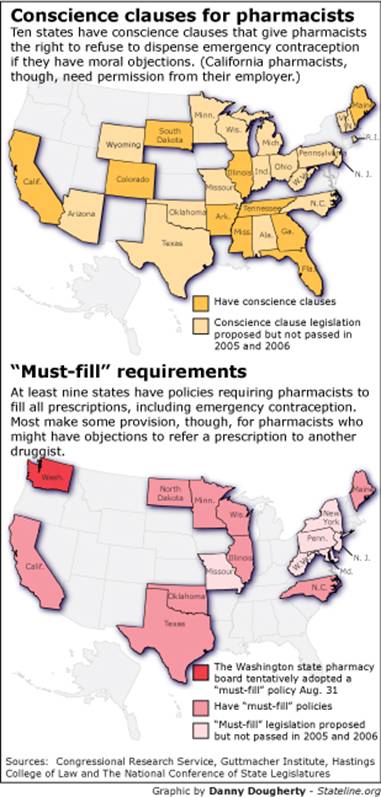
A multi-state issue
Like
Pharmacists have lost their jobs in
Massachusetts Gov. Mitt Romney (R), a possible presidential
contender for 2008, touched off a furor when his administration suggested that
Catholic hospitals would not be subject to a state law mandating that emergency
rooms offer emergency contraception to rape victims. He quickly reversed that
stance.
States are split over whether to give priority to health
care providers who have ethical concerns or to women seeking contraception.
Four states –
In addition,
The Washington Board of Pharmacy at first considered
enacting a conscience clause but reversed course after the governor weighed in.
If Gregoire's proposal receives final approval early
next year,
The American Medical Association passed a resolution last
year calling on pharmacists to fill all prescriptions. It even suggested that,
if no pharmacist within 30 miles of a patient would fill a script, the patient
should be able to buy the drug from the doctor instead.
New policy questions
The FDA's decision leaves states with even more questions to
resolve: Should stores be required to stock Plan B, now that it's a
non-prescription drug for women over age 18? Will pharmacy technicians, along
with druggists, be covered by conscience clauses? And should other states
follow the lead of nine states that currently let girls under 18 get the drug
without seeing a doctor?
These questions fall to the states because, while the FDA
regulates medicines, states police the doctors who prescribe drugs and the
pharmacists who dispense them.
In its long-delayed decision, the FDA on Aug. 24 agreed to
allow women age 18 and over to buy Plan B without a prescription, but kept the
prescription requirement for girls 17 and under. The manufacturer can sell the
drug only to stores and clinics where a pharmacist works. The medicine must be
kept behind the counter, and store employees must verify the age of the
purchaser. Men can pick up the drug, if they are old enough, for their
girlfriends, wives or other women.
When layered over existing state laws, the FDA rules create
new wrinkles.
Nine states already allow specially trained pharmacists to
give emergency contraception to patients who haven't visited a doctor.
Druggists are allowed to partner with doctors or to follow state regulations to
essentially write prescriptions for the drug on their own.
Under those arrangements, girls under age 18 in Alaska,
California, Hawaii, Maine, Massachusetts, New Hampshire, New Mexico, Vermont
and Washington may continue to get Plan B without seeing a physician.
The absence of a prescription requirement for women over 18
also could affect the already contentious debates over whether emergency rooms,
especially at Catholic hospitals, should be required to give the pill to rape
victims who desire it.
Seven states, including
Conscience clause issues also might be raised about other
pharmacy employees behind the counter. Lawmakers in
The FDA's unique arrangement that makes Plan B
non-prescription for women 18 and over but prescription for younger females
poses new policy questions in
Michael Patton, executive director of the Illinois
Pharmacists Association, said the FDA action could trump an administrative rule
imposed in April 2005 by Gov. Rod Blagojevich (D) that requires pharmacies that
stock other contraception to carry the morning-after pill.
"It's kind of like if I decided I didn't want to handle
Robitussin cough medicine. There is nothing in the law that would require me to
handle something that is not prescription-driven," he said.
But Sue Hofer, a spokeswoman for the Illinois Department of
Financial and Professional Regulation, which issued the rule, disagreed. Plan B
is still a prescription drug for girls, so the stocking requirements still will
apply, she said.
While
Since 1977,
Four pharmacists fired by Walgreen's last year for saying
they wouldn't dispense emergency contraception are challenging their
termination; their complaint is now with the Equal Employment Opportunity
Commission. Other pharmacy owners who worry they could lose their stores are in
court challenging the governor's rule mandating that they stock the
morning-after pill.
History of
conscience clauses
The controversy over women's access to Plan B has
parallels in the abortion debate, not just in principles but in tactics.
The introduction of Plan B as a prescription drug in 1999 –
and the manufacturer's application to the FDA in 2003 for over-the-counter
sales -- sparked a renewed interest in conscience clauses, which first cropped
up following the U.S. Supreme Court's 1973 Roe. v. Wade decision legalizing
abortion.
According to the Congressional Research Service, the first
conscience clause to become law was passed by Congress in 1973.
The so-called Church Amendment – named for U.S. Sen. Frank
Church, D-Idaho – prohibited officials from forcing federally funded
health-care providers to perform abortions. Since the mid-1990s, Congress has
passed numerous other restrictions to ensure that health-care professionals and
insurance programs aren't forced to participate in abortions.
Most recently, Congress began attaching a Hyde-Welden Amendment to appropriation bills in 2004. It bars
government agencies from treating health-care providers differently for
refusing to perform or pay for abortions.
States rapidly adopted conscience clauses – mostly for
abortion – after the Church Amendment.
Today, 46 states specifically give doctors the right to
refuse to perform abortions, according to the Guttmacher
Institute. By comparison, 16 states similarly shield doctors who object to
performing sterilization, and nine have laws allowing doctors to refuse to
prescribe contraception.
Plan B is distinct from the abortion pill RU-486. The abortion pill can terminate pregnancies for up to seven weeks, whereas emergency contraception will have no effect on an embryo that already has attached to the uterus. Pharmacists cannot dispense the abortion pill, because it must be taken in the presence of a doctor.