
Research

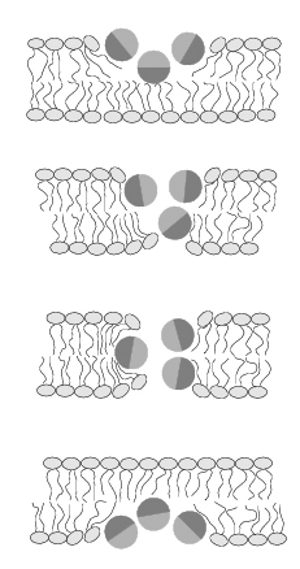
Membrane domains
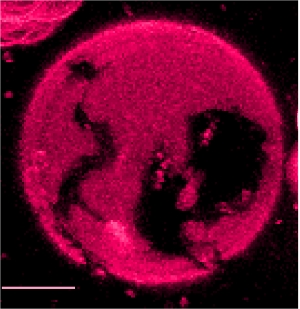
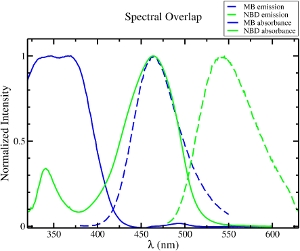
Mechanism of antimicrobial, cytolytic, and cell-penetrating peptides
Selected Publications
Pokorny, A., H. Birkbeck, and P.F.F. Almeida (2002). Mechanism and Kinetics of d-lysin interaction with phospholipid vesicles. Biochemistry 41, 11044-11056.
Pokorny, A., and Almeida P.F. (2004). Kinetics of dye efflux and lipid flip-flop induced by d-lysin in phosphatidylcholine vesicles and the mechanism of graded release by amphipathic, alpha-helical peptides. Biochemistry 43, 8846-8857.
Yandek, L.E., Pokorny, A., Floren, A.,
Knoelke, K., Langel, U., and Almeida, P.F.F (2007). Mechanism of the
cell-penetrating peptide transportan 10 permeation of lipid bilayers. Biophys J. 92, 2434-2444.
Gregory, S.M., Cavenaugh, A., Journigan, V., Pokorny,
A., and Almeida, P.F. (2008). A quantitative model for the all-or-none
permeabilization of phospholipid vesicles by the antimicrobial peptide cecropin A.
Biophys. J. 94, 1667-1680.
Almeida P.F.F. (2009) Thermodynamics of lipid interactions in complex bilayers.
Biochim Biophys Acta 1788, 72-85.
Gregory, S.M., A. Pokorny, and P.F.F. Almeida (2009) Magainin 2 revisited:
a test of the quantitative model for the all-or-none permeabilization of
phospholipid vesicles. Biophys. J. 96, 116-131.
Almeida, P.F, and A. Pokorny (2009) Mechanism of antimicrobial, cytolytic, and cell-penetrating peptides:
From kinetics to thermodynamics.
Biochemistry 48, 8083-8093.
Almeida, P.F. (2011) A simple thermodynamic model of the liquid-ordered state and the interactions between phospholipids and cholesterol. Biophys. J. 100:420-429
McKeown, A.N., Naro, J.L., Huskins, L.J., and Almeida, P.F. (2011) A thermodynamic approach to the mechanism of cell-penetrating peptides in model membranes. Biochemistry 50:654-662.
Clark, K.S., Svetlovics, J., McKeown, A.N., Huskins L, Almeida PF. (2011). What determines the activity of antimicrobial and cytolytic peptides in model membranes. Biochemistry 50:7919-7932.
Almeida, P.F., Best, A., and Hinderliter, A. (2011) Monte Carlo simulation of protein-induced lipid demixing in a membrane with interactions derived from experiment. Biophys J. 101:1930-1937.Almeida, P.F., Ladokhin, A.S., and White, S.H. (2012) Hydrogen-bond energetics drive helix formation in membrane interfaces. Biochim Biophys Acta. 1818:178-182.




| Where students went |



